
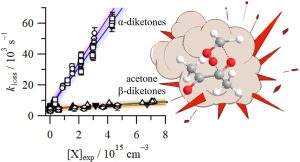
Our latest paper, exploring the photochemistry of acetaldehyde to form CH3 + HCO across a broad range of wavelengths, has been published in PCCP. We used a combination of time-resolved ion imaging, with nanosecond and picosecond pulsed lasers, and photofragment excitation action spectroscopy to identify three distinct dissociation mechanisms. At long wavelengths, dissociation occurs statistically on the S0 surface after many tens of nanoseconds. Dissociation at intermediate wavelengths is dominated by relatively fast dissociation on the T1 surface, leading to fast-moving CH3 radicals. At short wavelengths, a new pathway opens that is assigned to dissociation on the S0 surface, accessed via a conical intersection.
The article can be accessed here: 10.1039/c7cp02573d
This article was selected as part of the 2017 PCCP HOT Articles themed collection.

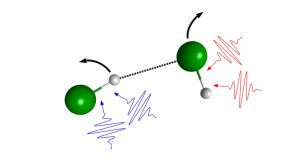
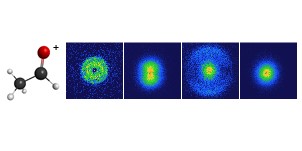



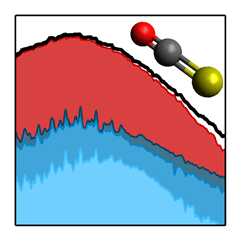 Our new paper published in J. Phys. Chem. A uses PHOtoFragment EXcitation (PHOFEX) spectroscopy to pick apart the first absorption band of OCS, the most abundant sulfur-containing molecule in the atmosphere. The PHOFEX spectra obtained probing electronically excited S(1D) and ground state S(3P) atoms are distinctly different; the former is broad and unstructured, while the latter shows distinct resonances that can be attributed to vibrational structure following direct excitation of a quasi-bound triplet state.
Our new paper published in J. Phys. Chem. A uses PHOtoFragment EXcitation (PHOFEX) spectroscopy to pick apart the first absorption band of OCS, the most abundant sulfur-containing molecule in the atmosphere. The PHOFEX spectra obtained probing electronically excited S(1D) and ground state S(3P) atoms are distinctly different; the former is broad and unstructured, while the latter shows distinct resonances that can be attributed to vibrational structure following direct excitation of a quasi-bound triplet state. 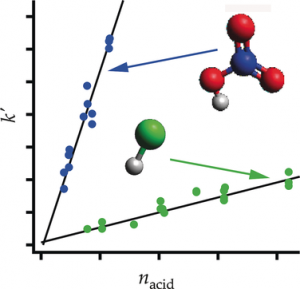
 The first is another collaborative paper with UCI colleague
The first is another collaborative paper with UCI colleague