Examining the Diverse Pathological Consequences of Microglial Absence
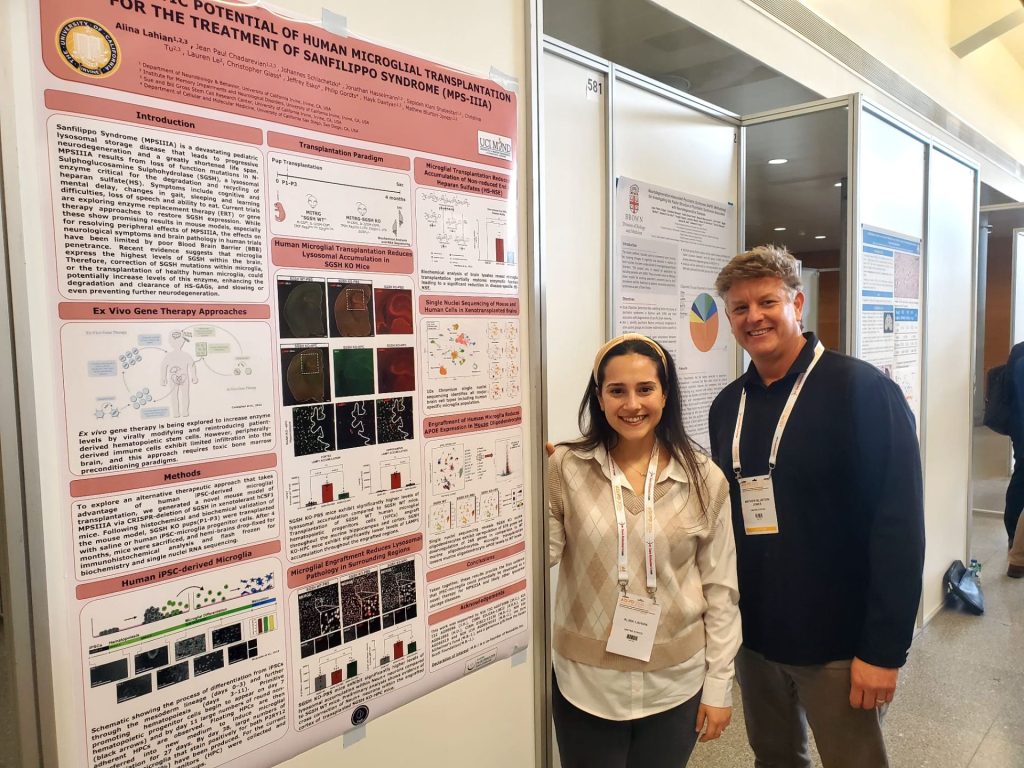
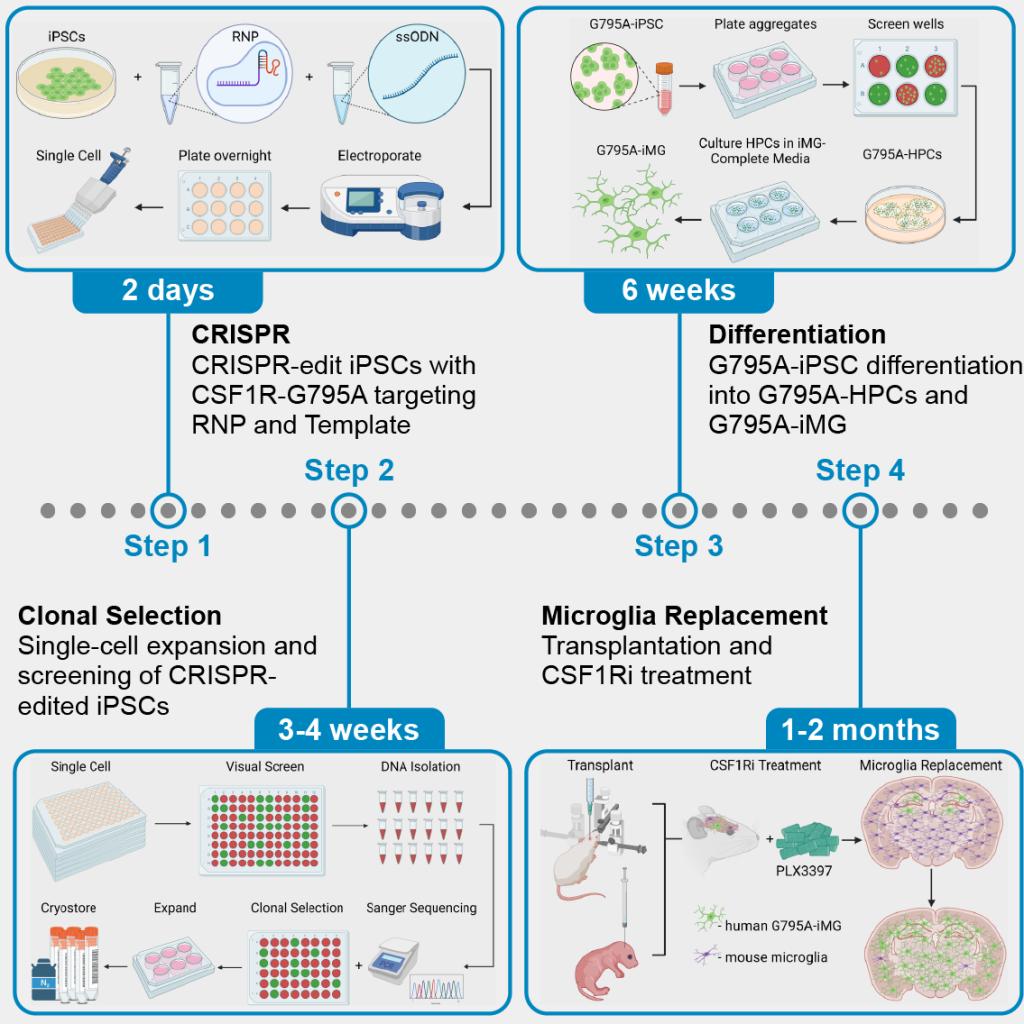
Our understanding of microglia, the principal immune cells of the central nervous system (CNS), continues to evolve as new models and approaches shift our perception of these intriguing cells. Previously envisioned as passive guardians of the brain, microglia have since been shown to play critical roles in development, neuroplasticity, and neurological disease.To further understand the role of microglia in AD and the impact of microglial absence on the aging brain, my thesis studies utilized ‘FIRE mice’, a genetic model that lacks microglia. FIRE mice harbor a homozygous deletion within the Fms intronic regulatory element (FIRE) super-enhancer, leading to a loss of CSF1R expression and congenital absence of microglia. To examine the role of microglia in AD pathogenesis, I crossed FIRE mice with 5xfAD mice that develop robust beta-amyloid plaque pathology. Remarkably, I found that absence of microglia promotes the development of cerebral amyloid angiopathy (CAA), brain calcification, and cerebral hemorrhages in AD mice. Importantly, transplantation of wildtype microglia prevents each of these pathological changes. To determine whether microglia absence alone can also induce pathological changes within the aging brain, I further examined 9-10 month-old FIRE mice in comparison to wildtype littermates and explored the impact of postnatal microglial transplantation, demonstrating that prolonged absence of microglia leads to the development of astrogliosis, calcification, and seizures, mimicking many of the pathological features of a rare human primary microgliopathy. Taken together, my thesis studies have revealed important roles for microglia in protecting the brain against age- and disease-related development of vascular, and white matter pathologies. These findings not only deepen our understanding of microglia’s roles in neurodegenerative diseases, but also provide initial evidence to support microglial transplantation as a viable therapeutic approach for a variety of neurodegenerative diseases.