The Role of the Hippocampus in Memory
The hippocampus is known to play a critical role in declarative memory, however the exact computations carried out in the hippocampus have been quite elusive. Over the past decade, empirical data across animals and humans has demonstrated that, consistent with computational models, the hippocampus is involved in pattern separation, the process of orthogonalizing similar experiences using distinct neural codes.
The hippocampal circuitry is complex with multiple input/output pathways. We are attempting to understand the different computational capacities in subregions of the hippocampus and the medial temporal lobe, as well as the information content processed and perhaps stored by each of these regions.
Our lab investigates the following areas:
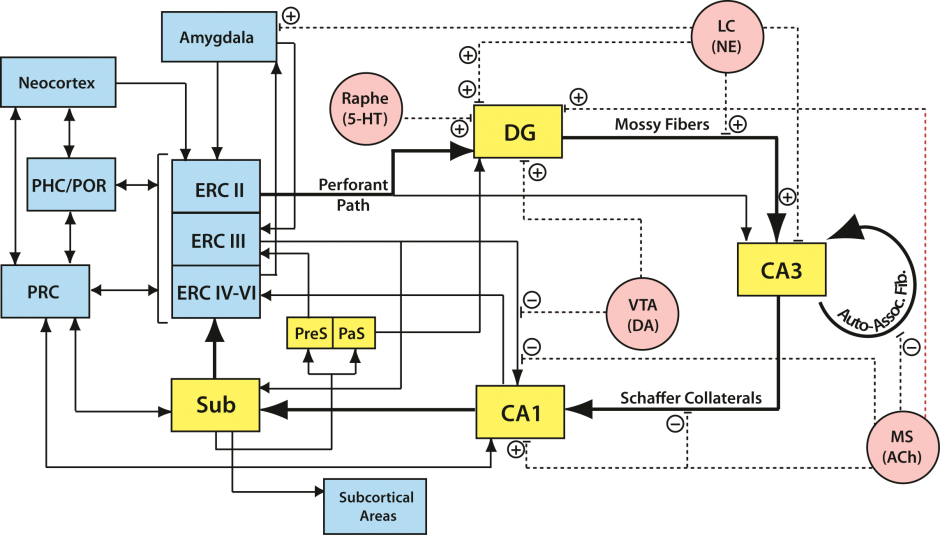
Episodic Memory Systems Involvement in Pattern Separation
The field of episodic memory research has been guided by decades of theory and computational models, which have proposed that episodic memories arise as a function of dynamic interactions between the hippocampus and the neocortex. The mechanisms by which these interactions manifest have remained elusive. Central to episodic memory formation and retrieval is pattern separation, the neural process enabling the storage and retrieval of similar yet distinct experiences. Using human intracranial recordings from the hippocamps and neocortex, we are identifying encoding and retrieval system dynamics that are conducive to pattern separation. We are finding that 4-5 Hz theta oscillatory dynamics underlying pattern separation during encoding and retrieval, both within and between the two systems. Theta power is recruited in both systems, and theta phase reveals underlying information transfer from the hippocampus to the neocortex during successful encoding, and from the neocortex to the hippocampus during accurate retrieval. This is consistent with extensive computational, behavioral and physiologic evidence for the role of theta as a fundamental rhythm reflecting a neural code in memory formation and retrieval.
Future Studies
Future projects will tease apart individual circuit components of the hippocampal-neocortical theta network. We will do so by characterizing how hippocampal subfields differentially interact with distinct neocortical modules (prefrontal, frontal, temporal, cingulate, and entorhinal cortices) and vice versa.
Impact
Our work unravels a mechanistic basis for the hypothesized episodic learning systems interactions. Future work building on this knowledge can develop theta-based therapeutic interventions for various conditions including addiction, post-traumatic stress, and age-related dementia.
Characterizing Hippocampal Computations
Numerous studies have demonstrated the level of decorrelation of hippocampal output compared to input patterns. However, a gap remains regarding the hippocampal computations facilitating such observed decorrelations. We therefore aim to characterize these computations using system identification, defining the system as a given hippocampal subfield. Using slice electrophysiology, we are currently estimating the hippocampal CA1 subfield transfer function (input-output mappings) by stimulating the CA3 shaffer collaterals and recording from the CA1 pyramidal cell layer.
Future Studies
We aim to characterize the transfer function for each of the hippocampal subfields. In doing so, we can link the constituent subfields together to generate a net hippocampal transfer function capturing computations occurring on neocortical input patterns, and their corresponding outputs back to the cortex.
Impact
Altogether, this is a region and hypothesis agnostic approach to identifying computations occurring in brain areas. In systematically applying this approach to different brain areas, and in linking these areas together, we can characterize how the brain computes, as a whole.
Domain-selectivity in MTL Cortices: Object vs. Spatial Pattern Separation
The hippocampus receives spatially segregated input from different subdivisions of the entorhinal cortex (medial – MEC and lateral – LEC), which in turn receive large inputs from the parahippocampal (PHC) and perirhinal (PRC) cortices respectively. The seminal discovery of grid cells in the MEC (Hafting et al., 2005) as well as the relative absence of spatially tuned cells in LEC reported by Hargreaves et al. (2005) has led to the proposal that entorhinal inputs to the hippocampus are organized as an extension of the PHC-PRC spatial-nonspatial dissociation. Evidence for this in humans has been elusive thus far. We have developed a novel task that simultaneously assesses object and spatial pattern separation, with the latter based on varying metric distances between the study and test items. Using high-resolution fMRI, we are currently testing the double-dissociation that object pattern separation will be selectively associated with PRC-LEC processing, while spatial pattern separation will be selectively associated with PHC/MEC processing. We further hypothesize that the DG/CA3 will not be selective to spatial vs. nonspatial information (i.e., domain-agnostic), while the CA1 may show selectively based on proximal vs. distal segments preferentially afferented by MEC vs. LEC respectively. Thus far, our data is highly consistent with a PRC/LEC vs. PHC/MEC division of labor, and furthermore with hyperdimensional pattern separation in the hippocampus. Our data suggests that some rudimentary form of pattern separation may be occurring in MTL cortices upstream of the hippocampus, and that ultimate orthogonalization is instantiated in the DG-CA3 network.
Future Studies
Impact
Temporal Processing
Time is an essential component of episodic memory, and recent studies have shown that hippocampal neurons are modulated not only by spatial location (i.e., place cells) but also by temporal duration (i.e., time cells; MacDonald et al. 2011). We propose that the DG plays a similar role in temporal pattern separation as has been proposed for spatial pattern separation. The function of temporal pattern separation is to allow the hippocampus to disambiguate information that is presented in temporal proximity. If hippocampal memory storage were modulated by time, then patterns of neural activity would change over time and produce less correlated temporally contiguous memories. We hypothesize that the dentate gyrus creates temporal signals that are reset by behaviorally relevant events and that encode time at a finer scale than the entorhinal inputs (i.e., temporal pattern separation). We are pursuing this possibility by using high-resolution fMRI during a novel temporal interference task that we developed.
Future Studies
Impact
Emotional Pattern Separation
It has been long recognized that emotional experiences are generally remembered better than neutral ones. In the past century, psychologists have provided a more nuanced perspective on this phenomenon showing that this is not entirely the case, and even more recently, neuroscientists have begun probing into the brain mechanisms underlying memory and emotion. While emotional arousal does improve the encoding of a memory, i.e., the more excited one is, the more likely one will remember an experience, it also narrows the focus of the memory to the emotional aspects of the experience. For example, when someone is shown an image of a person holding a weapon, they tend to fixate on the weapon rather than the other details in the image or the person’s face or scenery. This focus on the emotional aspects of a stimulus can further lead to overgeneralization errors.
In the lab, we have shown that subjects are less likely to discriminate images which are similar but unique if the image has an emotional valence. For instance, a subject is less likely to discriminate between two images of burning buildings than they are two images of pine trees. As described above, our fMRI studies have revealed that the activity of the dentate gyrus (DG) and CA3 of the hippocampus are involved in the discrimination of similar items; however, this activity as well as activity in the amygdala are modulated by negative images. Furthermore, in depressed subjects, DG and CA3 activity is reduced, while activity in the amygdala is heightened. In a subsequent study using intracranial EEG, we further elucidated this amygdala-hippocampus interaction showing that when stimuli are properly discriminated, these regions couple bidirectionally in the theta range; whereas, for improperly discriminated stimuli, this interaction shifts into the alpha range and is driven by the amygdala.
Future Studies
Impact
Emotional aspects of memories are generally emphasized over non-emotional details, and this can lead to overgeneralization errors — a fact that is often disregarded in eye-witness testimony. For negative emotions, the amygdala modulates the typical memory processes of the hippocampus. This modulation is greater for individuals with depression, which may provide some mechanistic account for the symptomology.
Project Team

Sandra Gattas
Graduate Student
